Read this K-value off the chart (approximately 21.3). In effect, it indicates the ease or difficulty of using distillation to separate the more volatile components from the less volatile components in a mixture. This quantity is widely used in designing large industrial distillation processes.

On the right-hand vertical axis, locate and mark the point containing the temperature 60☏.Empirical Correlation (dePriester Chart).

On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia. Example: does ethanol (1) and n-hexane (2) form an azeotrope.Example įor example, to find the K value of methane at 100 psia and 60 ☏. Many DePriester charts have been printed for simple hydrocarbons. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. These nomograms have two vertical coordinates, one for pressure, and another for temperature. This Demonstration applies a DePriester chart a set of nomograms to find the vaporliquid equilibrium ratio the gas phase mole fraction divided by the liquid. DePriester in an article in Chemical Engineering Progress in 1953.
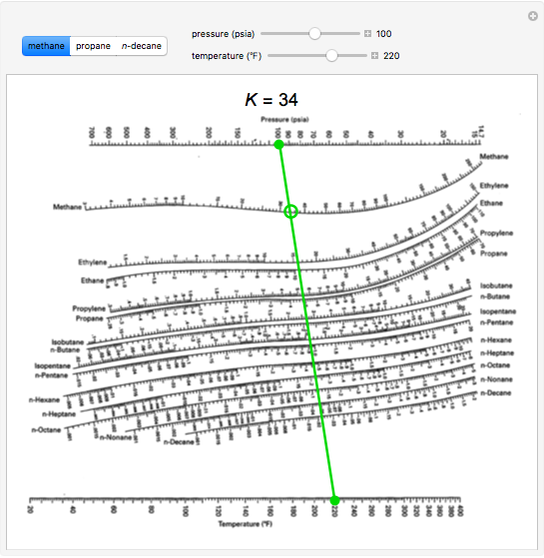
DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature. Use the DePriester chart to generate a temperature-composition diagram for isobutane and propane at 1000 kPa.
